Cellular morphogenesis is a fundamentally mechanical process in which many individual cells move and change shape, exerting forces upon and experiencing forces from their immediate neighbors, and in so doing they drive tissue-level deformations. None of an embryo’s cells know what the group as a whole is supposed to do or make. So how do they manage collectively to reorganize and deform themselves in a stereotyped way, producing just the right outcome every time?
In recent years, cell biologists have begun to uncover and characterize the machinery that individual cells use to generate local contractile or protrusive forces, to polarize, to establish and transmit forces through local adhesive contacts with one another or with extracellular substrata, and so on. This core machinery turns out to be strikingly conserved within the animal kingdom.
Given this conservation, it seems likely that same core machinery that propels isolated cells in vitro also drives the movements of embryonic cells during morphogenesis. The fundamental question then becomes: How does conserved cytoskeletal machinery operating within each of many cells to generate purely local forces, cause a population of cells, whose members are interlinked by adhesive machinery, to reorganize and deform in a characteristic pattern? How do differently fated embryonic cells regulate the local force-generating and adhesive machinery differently so that they collectively produce a stereotyped global outcome? How has the evolutionary process tuned this machinery to produce morphogenetic variation across phylogeny?
To address these questions, requires comprehending how local force-generating processes operating at cellular or subcellular levels yield tissue-level consequences. Physical law dictates that, at every moment, forces arising locally anywhere within an embryo resolve themselves simultaneously and globally into instantaneous patterns of cell movement and deformation. It is often difficult to intuit the outcome of this resolution. At the CCD, we combine detailed computer simulation models with modern imaging tools, micromanipulation, and molecular perturbation, to approach a mechanistic understanding of how some specific morphogenetic machines actually work.
Our recent work has focused on understanding the morphogenetic machine that drives the formation and extension of the notochord, and in related work, of endoderm invagination, and for both studies we avail ourselves of the many advantages of ascidian embryos. The ascidian notochord forms from a simple monolayer sheet of forty cells that reorganize themselves in about 6 hours without cell divisions to form an extended rod of coin-shaped cells (Munro and Odell 2002a&b).
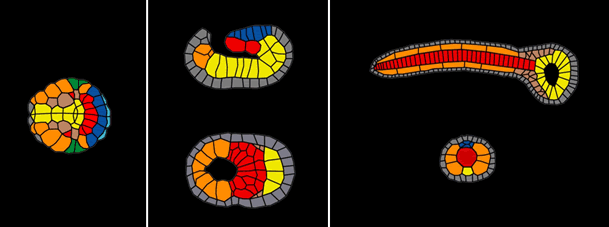
Schematic view of different stages of notochord formation in an ascidian embryo. Left panel: early gastrulation; Middle panel: late gastrulation; Right panel: tailbud stage. Presumptive notochord cells are shown in red, endoderm yellow, muscle orange, epidermis gray, and neural plate blue.
This transformation involves two simultaneously occurring processes: invagination of the notochord plate forms a cylindrical rod; and intercalation of cells across the plate and about the circumference of the rod causes it to become longer and thinner.
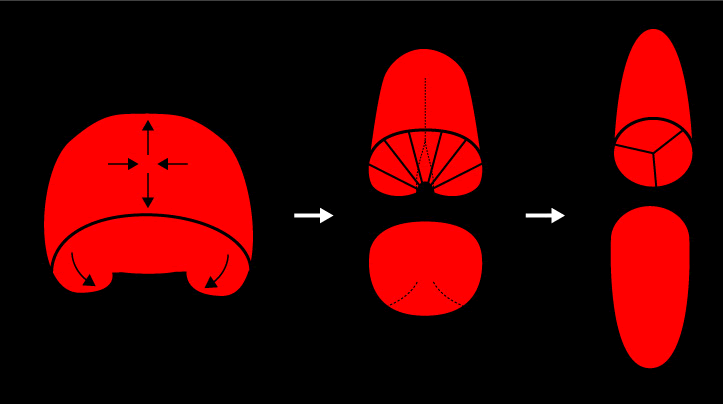
Schematic view of the two morphogenetic prcesses that simultaneously transform the notochord plate into a cylindircal rod.
During the 6 hours in which these events occur (and not before or after), individual notochord cells crawl actively across the basolateral surfaces of their notochord neighbors. This crawling appears to be polarized with respect to both the axis of notochord extension and the apico-basal axis of individual notochord cells: cells appear to crawl more actively along the axis of intercalation (perpendicular to the axis of notochord extension) and they appear to crawl more actively near the basal surface of the notochord. These observations suggest that active local motility, expressed identically by all notochord cells and polarized with respect to the apico-basal and AP axes of the notochord plate, could generate the asymmetric global distribution of forces necessary to cause the invagination of the notochord plate on the one hand, and the convergent extension on the other.
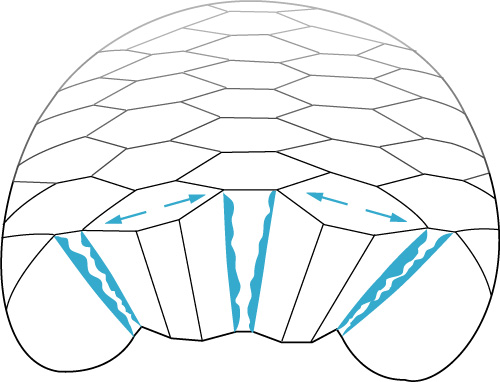
Notochord cells converge and extend by crawling locally across the basolateral surfaces of their adjacent neighbors in much the same way that an isolated fibroblast would crawl across an external adhesive substratum.
Based on these observations, we hypothesized that the same conserved cytoskeletal and adhesive machinery that endows a single isolated cell with the ability to crawl across an isolated adhesive substratum could account, both qualitatively and quantitatively, for the ability of a sheet of the same cells to converge and extend or invaginate. To see whether and how this might be true, we built a computational framework to predict, from specific assumptions on the conserved contractile, protrusive and adhesive mechanics operating locally within each of many cells, what a sheet of such cells would do. The basic idea was build individual model cells from a collection of discrete elements- each representing a small piece of the cell cortex or internal cytoplasm, the nucleus, adhesion proteins, and so on- and to endow each of these discrete elements with local kinetic and mechanical properties designed to mimic those measured empirically for their real counterparts. We wanted to build in only the local behaviors and let the global consequences emerge. To model a sheet of cells, we simply arrange many model cells as neighbors and then allow them to adhere.
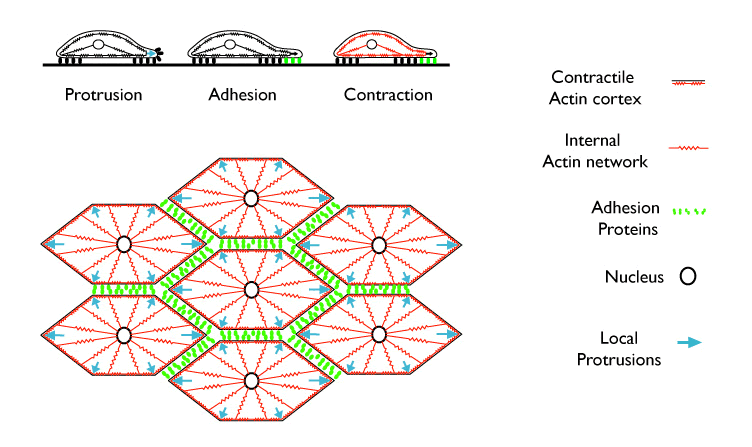
The top tryptich shows how local protrusive, contractile and adhesive machinery might operate within an individual cell crawling across an isolated substratum. Below, a hypothetical view of how the same machinery might operate within a monolayer sheet of embryonic cells during convergent extension.
Initially, we focused on three fundamental cytomechanical behaviors, thought to be necessary for the directed movements of single cells across isolated substrata (e.g. Alberts et al 2003, Bray 2000): 1) localized actin-dependent protrusive extension of the leading edge; 2) heterophilic or homophilic adhesion mediated by reversible binding between proteins located on adjacent cell surfaces and dynamically anchored to the underlying cortex; and 3) actomyosin-based contraction of the cortex. When we endowed model cells with these three local behaviors, tuned parameters governing contractile, adhesive and protrusive mechanics to match empirical measurements, and biased protrusive extension to a single “leading edge”, we found that a single model cell would crawl in a realistic way across a rigid external adhesive boundary. Thus the textbook model of how individual cells crawl works mechanically.
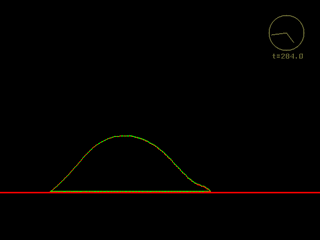
|
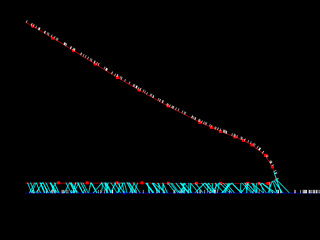
|
Click on the panels above to see the QuickTime movies of a simulated cell crawling across a flat adhesive substratum. Right: High resolution view. [new window 700 x 660 pixels, 2.2 MB]
To our surprise, we found that a monolayer sheet of the same cells, identically tuned, with protrusive extension biased medially and laterally, would converge and extend! Moreover, the basic convergent extension behavior was remarkably robust with respect to quantitative variation of parameters governing protrusive, contractile and adhesive mechanics.
 |
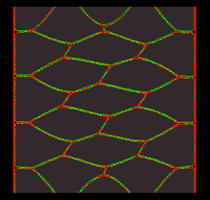 |
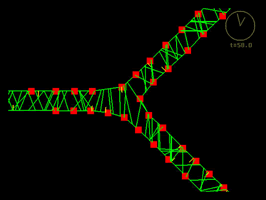 |
Click on each of the panels above to see the timelapse movie. Left: The real thing, filmed within an intact embryo [opens in new window 360 x 360 pixels, 3.7 MB]; Middle: Model simulation showing how a sheet of model cells executes convergent extension [opens in new window 680 x 630 pixels, 1.4 MB]; Right: A close up view of one three cell junction from the same simulation [opens in new window 680 x 560 pixels, 1.5 MB].
Munro and Odell (2002a) Polarized basolateral cell motility underlies invagination and convergent extension of the ascidian notochord. Development 129, 13-24.
Munro and Odell (2002b) Morphogenetic pattern formation during ascidian notochord formation is regulative and highly robust. Development 129, 1-12.
Wallingford, JB, Fraser, SE, and Harland, RM. (2002) Convergent extension: the molecular control of polarized cell movement during embryonic development. Developmental Cell 2(6), 695-706.
Keys DN, Levine M, Harland RM, Wallingford JB. (2002) Control of intercalation is cell-autonomous in the notochord of Ciona intestinalis. Developmental Biology 246(2), 329-40.