von Dassow, Bement, Foe, Odell
Despite no lack of attention from cell biologists, geneticists, and even theoreticians, animal cell cytokinesis — during which the cell actually pinches in two following chromosome segregation — remains an unsolved mystery. It is widely accepted that progress of the division furrow is based on the contraction of a bundle of aligned actin filaments and myosins, the contractile ring, which is a remarkably conserved feature of dividing animal cells. Also, classical experiments show that animal cells establish the furrow region in relation to the position and orientation of the microtubule-based mitotic apparatus: cells of various types correct for experimental displacement of the metaphase spindle due to centrifugation, deformation, or micromanipulation, managing still to cleave such that the furrow separates one set of chromosomes and spindle pole from the other set.
|
|
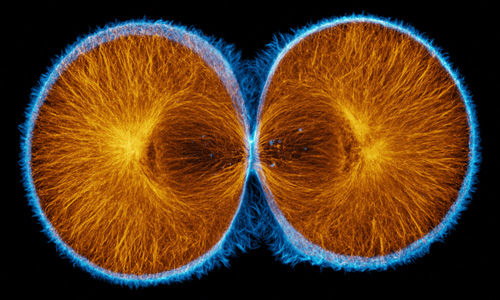
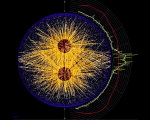
Actin (blue) and microtubules (orange) at the end of cytokinesis in a green urchin zygote. |
What is not clear is how the spatial information immanent within the microtubule-based mitotic apparatus, deep in the cell, coordinates with the cell cortex to direct formation of the contractile ring and the initiation of furrowing at the appropriate site (see picture below). Because of the uniformity of the contractile ring, many biologists seem to have assumed that all animal cells share a common mechanism for furrow positioning and contractile ring assembly. However, animal cells are diverse in size, shape, and other properties. Classical experiments implicate the astral microtubules, but more recent work points toward the "central spindle" and even to factors localized to the chromosomes. This apparent conflict between classical and recent results says that either animal cells follow widely different rules for furrow specification (not unlikely), or that cells do something more subtle than simply reading off one dominant spatial cue (also not unlikely).
The nature of the stimulus conveyed from the mitotic apparatus to the cortex remained obscure until recently. Recent work in several labs, including ours, identifies the small GTPase Rho as a key link. Rho, which has well-characterized roles in myosin activation and actin assembly, is required for cytokinesis in virtually all animal cells where it has been tested. Many regulators of Rho have been uncovered in genetic screens for cytokinesis defects. We showed, using a probe for Rho activity in live cells, that active Rho accumulates in a narrowly-defined equatorial zone immediately before the onset of furrowing. This signaling molecule has become the prime candidate for the physiological signal linking microtubule geometry to contractile protein assembly in the cell cortex during cytokinesis.
While it may be that most animal cells use many of the same molecular components to accomplish division, they may not all use that machinery in the same way. Although many of the classical descriptive and experimental studies on furrow induction were performed on embryos of sea urchins and other marine invertebrates, most of the recent molecular biology of cytokinesis comes from studies on Drosophila and C. elegans or on cultured mammalian cells. Nevertheless, hypotheses about cytokinesis continue to be dominated by observations that pre-date molecular probes and confocal microscopy. One of our major research projects is therefore to re-examine cytokinesis in echinoderm embryos using state-of-the-art microscopy and molecular probes, to find out how to reconcile classical observations with emerging knowledge of the molecular details of cell mechanics. Our current efforts focus on the dynamics of the cytoskeleton during cytokinesis, and on the mechanism of Rho activation in the cleavage furrow.
In the past many studies on cytokinesis were interpreted in the context of a debate between "polar relaxation" and "equatorial stimulation" models for the furrowing stimulus. The key points of debate were, when do microtubules reach different regions of the cell cortex, and what do they do when they get there? Do they relax the cortex, or promote contractility? This is still unresolved because microscopists consistently find that astral microtubules reach the polar cortex first and in greater numbers, whereas many experiments imply that microtubules promote cortical contractility.
|
|
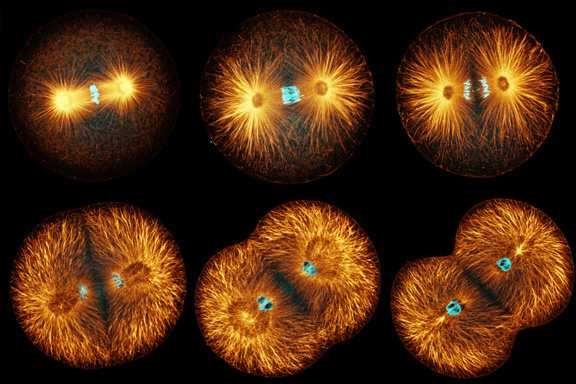
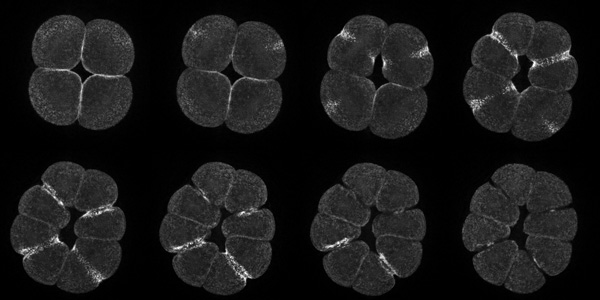
Sand dollar zygotes during first mitosis, fixed and stained for microtubules and chromosomes (single confocal sections). Top row: metaphase, early anaphase, late anaphase; bottom row: successive stages in furrowing. A variety of experiments imply that microtubules stimulate cortical contractility in the cell equator, but also many experiments purport to show that microtubules are dispensable after mid-anaphase (right-most frame in the top row). In an urchin egg like this one, microtubules have reached the cell poles at this stage, but not the equator. They only reach the equator after furrow initiation (left frame, bottom row). How can this be? |
This debate seems to overlook two important possibilities: first, maybe the microtubules do many things at once; second, maybe not all the microtubules are equivalent. In any case, neither polar relaxation nor equatorial stimulation constitute a satisfactory, all-encompassing explanation for many variants of cytokinesis, or cytokinesis-like processes such as somatic budding in flies, unequal cleavage in echinoid and other embryos, polar lobe formation in molluscs and annelids, etc. Again, perhaps cells have more than one trick in their bag when it comes to such a fundamentally important process as division.
In earlier work on syncytial Drosophila embryos, which do not undergo cytokinesis, we described 1) a cytoplasmic actin network that comes and goes with each mitotic cycle during nuclear migration, and 2) evidence for active re-organization of actin and myosin by astral microtubules. We discovered a very similar network of actin filament bundles in early sea urchin embryos (among other species), which appears and disappears in phase with mitosis, and whose non-uniform distribution is somehow dependent on astral microtubules.
|
|
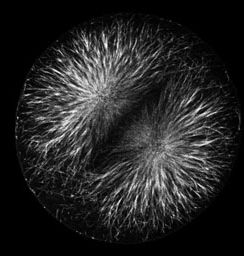
|
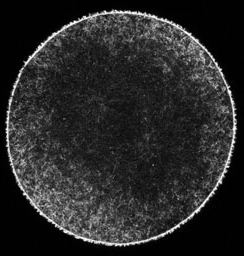
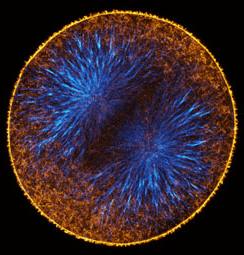
Actin filaments (left) and microtubules (right) in a late anaphase sand dollar zygote, 0.5 micron confocal section. To view a fly-through of the entire stack, choose a small version [QuickTime movie v6.2 or later, 3.3 MB] or a slightly larger version [QuickTime, 7.7 MB].
We're currently working to visualize actin and microtubules in live embryos to find out whether transport, i.e. along microtubules, brings contractile material to the equator. Preliminary results are that this does not pertain in urchin embryonic cells; the actin in the cleavage furrow seems to appear in place, implying local assembly, not transport. We are also investigating recruitment of myosin II to the furrow region by staining embryos with antibodies specific for the serine-19 phosphorylated myosin II regulatory light chain ("activated" myosin). During normal cell division, phosphorylated myosin is present throughout the cortex in interphase through metaphase, but leaves the cortex in early anaphase, only to return specifically to the equator in late anaphase. This too seems consistent with local recruitment rather than transport of pre-existing contractile material: there simply isn't any active myosin II on the cortex just before the furrow appears.
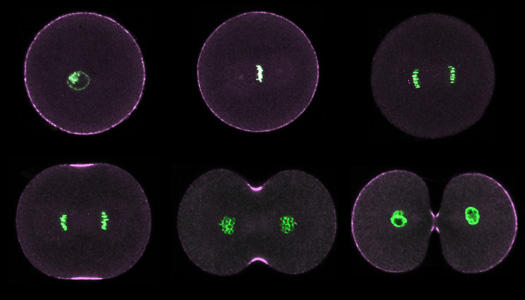
|
|
Purple urchin zygotes during first mitosis, fixed and stained for DNA (green) and phosphorylated myosin II (magenta; single confocal sections). Top row: interphase, metaphase, anaphase. Bottom row: early, middle and late telophase. |
In the course of these studies we also found that microtubules may be involved for a longer period in cleavage than previous reports imply. It was once widely believed, on the basis of experiments using microtubule poisons like nocodazole, that microtubules become dispensible for furrowing shortly after the metaphase/anaphase transition, even though the furrow does not begin to ingress until after anaphase is complete. This conclusion, however, does not accord with many experiments showing that the furrow can "correct" or even re-form entirely if the mitotic apparatus moves after furrow initiation.
The resolution to this particular conflict is both simple and profound: not all astral microtubules are the same. We found that treatment with high doses of nocodazole eliminates all of the astral microtubules very rapidly in interphase, prophase, and metaphase, but not anaphase or telophase. During anaphase and telophase, some astral microtubules are resistant to nocodazole-induced disassembly, and in three species of echinoid zygote, these stable astral microtubules predominantly point toward the furrow region. Indeed, the ends of stable microtubules correlate well with the initial spots of myosin II recruitment to the furrow zone. How these stable microtubules form, and whether they are in fact required for furrow induction, is one of the current focuses of our research.
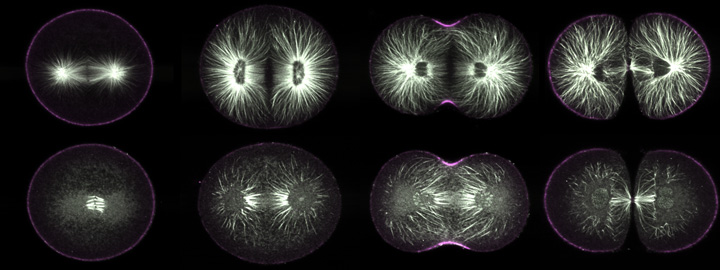
|
|
Purple urchin zygotes during first mitosis, fixed and stained for microtubules (white) and phosphorylated myosin II (magenta; single confocal sections). Top row: metaphase, anaphase, telophase, and furrowing completed, in untreated cells. Bottom row: zygotes of equivalent stages, but treated five minutes before fixation with nocodazole. In metaphase, only kinetochore fibers survive nocodazole treatment; the astral microtubules all disappear. In anaphase, however, some astral microtubules remain, and during telophase it is apparent that most of these point toward the equator (third panel, bottom row). |
Meanwhile, the rest of the astral microtubules - the dynamic ones, which disappear rapidly after nocodazole treatment - also have a role in defining the furrow zone. In cells treated with nocodazole after anaphase onset, the equatorial myosin zone is wider than normal; in cells treated just after the metaphase/anaphase transition, the myosin recruitment zone even expands to cover the whole cell. Thus dynamic astral microtubules, during anaphase, delimit the region of the cortex where myosin recruitment takes place.
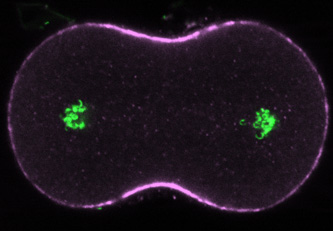
|
|
Ten minutes after treatment with nocodazole in late anaphase, the equatorial myosin zone and the furrow itself are much broader than normal, reflecting a continuing role for dynamic microtubules in shaping the domain of contractile protein recruitment during telophase. |
Our current working hypothesis is that stable microtubules and dynamic microtubules have opposite effects on the delivery to the cortex of Rho regulators (see below). The stable microtubules, by virtue of their persistence, ought to make better roads than dynamic microtubules, which might depolymerize from beneath motor-driven cargos in transit. However, because dynamic microtubules would still bind to available motors, they would soak up the available motor-associated Rho regulators, thus keeping them from being delivered to the cortex. We are using a newly-developed computer simulation to test whether this is a plausible and robust mechanism for positioning the cleavage furrow.
| 140.7 MB [675 x 640 pixels] |
MKLP1 motors on microtubules in urchin egg(Garrett Odell & Victoria Foe)
We discovered that the small GTPase Rho is activated in a narrow zone that immediately prefigures furrow formation in both urchin and frog embryo blastomeres. Our work follows recent discoveries implicating Rho and its regulators in either the stimulation or maintenance of cytokinetic furrowing in diverse cell types, including mammalian cultured cells, Drosophila embryonic and cultured cells, and C. elegans embryos. Mabuchi and colleagues (Zygote 1:325-331) showed more than a decade ago that injection of C3 exoenzyme, a bacterial toxin which inactivates Rho, into sand dollar eggs prevents them from cleaving, without interfering with mitosis. Click on the figure below to see a time-lapse movie of sand dollar eggs injected with C3:
We used a probe for active RhoA developed in the Bement lab. This probe fuses eGFP to the Rho-binding domain of rhotekin. The rhotekin RBD binds specifically to active, GTP-bound Rho, and is the basis of commercially-available Rho activity assays. The probe is synthesized from injected mRNA, and when not bound to active Rho, diffuses throughout the cytoplasm. However, sites where active Rho is concentrated accumulate the probe by diffusion entrapment. We found that immediately prior to furrowing, active RhoA appears in a tight equatorial band.
|
|
Projections of 18 consecutive 1-micron sections through a live eight-cell purple urchin embryo expressing GFP fused to the rhotekin Rho-binding domain. These were deconvolved from the first movie in the gallery below. |
RhoA is active throughout furrowing, and disappears once the furrow is fully ingressed. The zone of Rho activity superficially matches the zone of myosin recruitment, and since all cortical myosin recruitment is abolished in C3-injected cells, Rho is causally upstream of contractile ring assembly. We also found that the Rho zone width remains roughly constant throughout furrow ingression, but scales linearly with cell diameter. For details, please consult Bement, Benink and von Dassow (2005) JCB 170:91-101. The movies here accompany that paper.
We found, furthermore, that the formation of the equatorial RhoA activity zone is independent of actin assembly; that is, it's not a consequence of furrowing. Also, we noted a curious phenomenon in cytochalasin-treated cells. As the cortex loses its stiffness, slender tubular invaginations of the plasma membrane intrude toward the spindle poles in a microtubule-dependent way. This bizarre "lightning" may reflect a microtubule-dependent removal of active Rho from the cortex, a possibility we will explore in the future.
Using physical manipulation of cells, we showed that the Rho zone is shaped by the proximity of the mitotic apparatus to the cortex, and responds rapidly to changes in the position of the spindle mid-zone.
In short, Rho signaling is at heart of the physiological mechanism by which the geometry of microtubules is coupled to the formation of the acto-myosin contractile ring. Specifically, we believe, based on its known function in other cells and on experiments we are presently conducting, that Rho activation at the equator is based on delivery of Rho regulators by astral microtubules; that increased availability of active Rho leads to recruitment of myosin II; and that the furrow is actively maintained throughout cleavage by delivery of Rho regulators, both positive and negative.
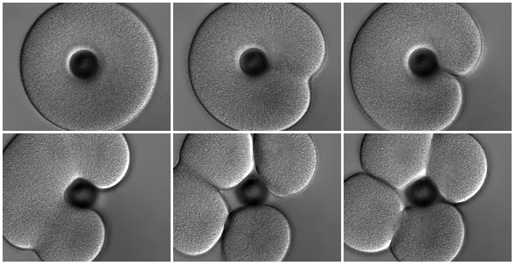
As we mentioned above, most of the classical experiments that define how the furrow forms in response to the mitotic apparatus were performed in urchin eggs. The principle conclusion, stemming chiefly from the work of Rappaport and Hiramoto, was that the juxtaposition of two astral microtubule arrays suffices to induce furrowing, and that neither the spindle itself nor the chromosomes is essential for furrow formation. The best-known demonstration of this conclusion is Rappaport's creation of a toroidal sand dollar egg. At first cleavage the toroidal cell develops a complete furrow across one side, bisecting the mitotic apparatus to yield a binucleate, U-shaped cell. In the second cleavage, furrows cross each of the two mitotic spindles, but another "secondary" furrow crosses between the two spindles, where there are no chromosomes or spindle fibers, thus cleaving the U-shaped cell into four.
|
|
A toroidal sand dollar zygote conducting the first two cleavage divisions; this is a repeat of Rappaport's famous experiment. Top row: at first cleavage the cell makes one furrow crossing the single spindle. Bottom row: at second cleavage the cell mounts furrows across both spindles (left panel) and then a "secondary" furrow forms between the two spindles (middle panel). Click here to see the movie (12.8 MB). |
However, it has been unclear to what extent these conclusions apply to other cells. Much of the recent work on genetically-tractable model organisms implicates proteins associated with the central spindle or the chromosomes in either furrow induction, furrow maintenance, or cytokinesis. Also, in a direct test of whether Rappaport's conclusions are general, Cao and Wang showed that in cultured mammalian cells, the furrow forms near the central spindle, not at the site where astral microtubules are juxtaposed, and they concluded that the central spindle is the locus of furrow induction in cultured cells. Nevertheless, there is strong evidence in C. elegans embryos that astral microtubules influence furrow position, even if the central spindle also plays an important role.
We decided to find out whether C. elegans embryos actually behave the same as urchin eggs, using experiments directly analogous to several of the classical urchin manipulations. By perforating zygotes or one of two cells, we showed that furrows form both where asters overlap and also where the central spindle comes close to the cortex. However, only furrows that cross the central spindle are likely to complete.
We also discovered we could fuse cells with a UV laser, thus creating binucleate cells. In more than half of these cells, the two nuclei migrate together, and a tetrapolar spindle forms. But if the two nuclei remain separate, they develop two independent spindles that are oriented in parallel to each other. Furrows form not only across these spindles but also between them.
Next we found we could make anucleate, centrosome-containing cells by severing one pole from the spindle at metaphase. Anucleate cells result when the furrow fails to pass between the two sets of sister chromosomes after anaphase. Invariably, the severed centrosome in an anucleate cell duplicates, swells as the "cell" enters M-phase, and the two daughter centrosomes take up opposite positions in the cell, even rotating into position in the posterior lineage, as the spindle normally would. Also invariably, deeply-ingressing furrows appear between the two centrosomes in anucleate cells. Most do not complete, but regress around the time abscission would normally take place. The anucleate cells go through several rounds of centrosome replication and attempted cleavage.
These results are almost isomorphic to the classical results on sea urchins, except that in worms it appears that furrows need to cross the central spindle to complete. But what does the central spindle have to do with furrow progression? We looked at the behavior of perforated cells deficient for Spd-1, without which cells lack a central spindle. We were surprised to find that Spd-1-deficient cells appear to be less selective than wild-type cells about which furrow will survive: aster-induced furrows ingress deeper and may even complete in perforated, Spd-1 deficient cells.
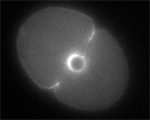
| 1.9 MB |
Two furrows complete in Spd-1-deficient, perforated zygote. (Jalal Baruni & George von Dassow)
This leads us to suggest that perhaps the central spindle, as the biggest group of bundled, anti-parallel microtubules, hoards some factor that is normally required for furrow progression (likely the centralspindlin complex), and that furrows which are aimed across the central spindle have access to this hoard, whereas furrows aimed elsewhere do not. Thus, in effect, the central spindle "ratifies" furrows that are aimed correctly. This work is described in Baruni, Munro, and von Dassow (2008) J Cell Science 121:306-16
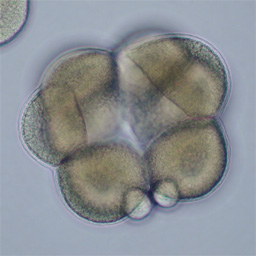
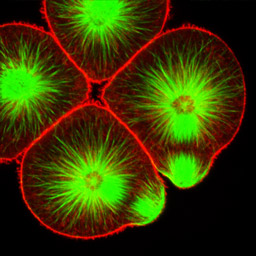
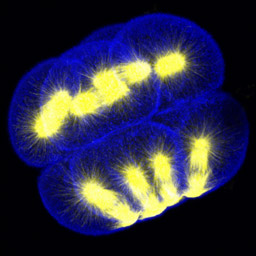
At fourth cleavage, echinoid embryos undergo a highly unequal division in the vegetal cells. The daughters of the vegetal four cells in the eight-cell embryo differ by approximately 30-fold in volume, such that the 16-cell embryo consists of eight equal-sized animal-pole cells (the mesomeres), four large macromeres in the vegetal half of the embryo, and four tiny micromeres at the very vegetal pole. Unequal cleavage is based on the eccentric positioning of the mitotic spindle, and subsequent stimulation of furrowing around just a tiny bud that becomes the micromere.
|
|
|
Left: 16-cell green urchin embryo, animal pole up and vegetal pole down; Middle: Single confocal section of an 8-cell sand dollar embryo initiating fourth cleavage, stained for tubulin (green) and actin (red); Right: projection of confocal Z-series through an 8-cell purple urchin embryo in metaphase, stained for tubulin (yellow) and actin (blue), animal pole up and vegetal pole down. Click on the right panel to view it as a 3D movie [QuickTime, 397 K].
We are interested in this process both because it presents an opportunity to investigate spindle orientation and microtubule/cortex interactions, and also because the asymmetric furrow defies easy exaplanation by either polar relaxation or equatorial stimulation. If the contractile ring formed as a consequence merely as a consequence of relaxation of the cortex by astral microtubules, why wouldn't the aster associated with the future micromere cortex stimulate capping of cortical actin and myosin at the opposite end of the cell? Instead the furrow overlaps the distal tips of the vegetal aster's microtubules. It is equally difficult to imagine that furrowing requires microtubules from adjacent asters to meet at the equator, because the other spindle pole is located nearly in the center of the cell, and astral microtubules from this pole barely reach the cortex anywhere until telophase.
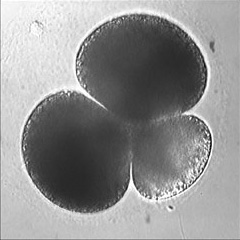
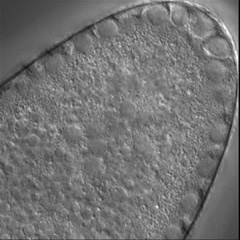
Invertebrate embryology provides a wealth of variations on the theme of cytokinesis. We are interested to explore some of these natural experiments to figure out how cells adapt the cleavage mechanism to different tasks and cellular properties. Some of the most interesting such phenomena are the unequal fourth cleavage in urchins (discussed above), unilateral cleavage in jellyfish, polar lobe formation in some molluscs and annelids, as well as pseudocleavage furrowing in insect embryos. Although the latter process is familiar and well-studied, for many of these cases there has been very little basic descriptive work on the configuration of the cytoskeleton during cleavage: for instance, in jellyfish embryos that cleave from one side of the cell only, is the contractile ring complete? And if so, why doesn't the cell surface furrow on one side; is there something in the way? Or if the ring is not complete, how does it drive the furrow in at all? Someone ought to take a look...
|
|
|
Left: the jellyfish Aequorea halfway through first cleavage; Middle: the scaphopod Pulsellum at the "trefoil" stage; Right: the wasp Nasonia at the syncytial blastoderm stage. View a time-lapse movie showing early cleavage in a variety of different embryos [QuickTime, 28.5 MB].